Offering scalable quality grades from Research to GMP, PlasmidFactory supports advanced gene-based innovations worldwide.
Minicircle DNA – The Core Technology for Next-Generation Therapies
PlasmidFactory’s patented Minicircle DNA technology removes all bacterial backbone sequences, leaving only the therapeutic expression cassette in a small, supercoiled DNA molecule.
This minimalistic vector design enables:
-Reduced immunogenicity and lower DNA toxicity
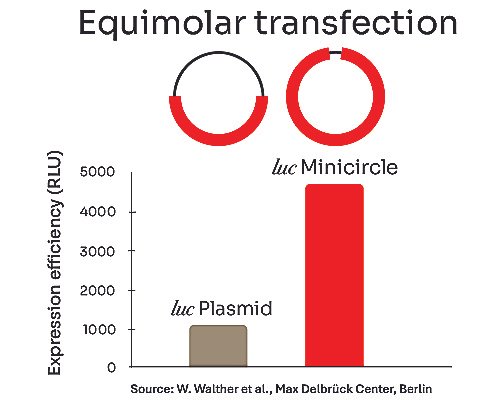
-Higher transfection efficiency and improved cell viability
-Stronger, more stable gene expression
-Backbone-free design available up to GMP Grade for safer, regulatory-aligned manufacturing

Minicircle DNA provides clear advantages across advanced applications. It enhances virus-free cell engineering such as CAR-T, CAR-NK, and HSPCs by improving transfection, expression, and viability while enabling efficient non-viral gene transfer — with clinical programs already underway. In viral vector production, its backbone-free structure prevents retro-packaging of bacterial sequences, ensuring vector purity, functional yield, and safety. As a template for IVT and vaccination platforms, Minicircles support cleaner and more reproducible mRNA manufacturing workflows.
Whether you are developing CAR-T, CAR-NK, viral vector strategies, RNA therapeutics, or novel in vivo gene delivery approaches — PlasmidFactory delivers the clean, reliable DNA foundation your program depends on.
PlasmidFactory – The Better Way to DNA.
www.plasmidfactory.com
Accelerate your manufacturing with Minicircle DNA.
https://www.plasmidfactory.com/custom-dna/minicircle-dna/
Webinar: Why DNA Quality Matters
March 26, 2026 | 17:00 CET / 16:00 GMT / 11:00 EST / 08:00 PST
https://www.plasmidfactory.com/events/why-dna-quality-matters-from-research-to-gmp/




