






























Eli Lilly Japan K.K. has decided to invest 20 billion yen in its Seishin Plant (Kobe City, Hyogo Prefecture),
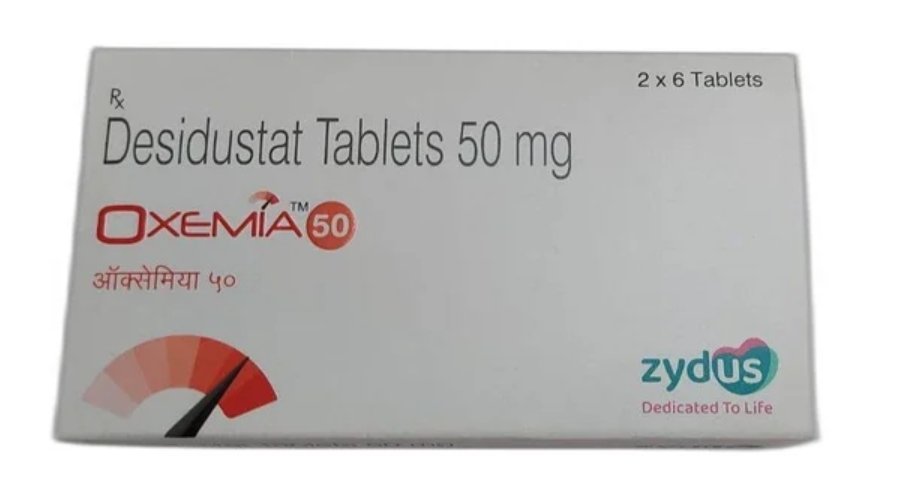
India-based Zydus Lifesciences has announced that its innovative drug Desidustat tablets, licensed to the subs

Aicuris Anti-infective Cures AG has entered into a definitive agreement under which Japan's Asahi Kasei,

China-based startup Excalipoint Therapeutics, a biotechnology company developing next-generation T-cell engage

Taiwan-based Medigen Vaccine Biologics Corp. (MVC) and Substipharm Biologics, based in Paris, have achieved a
Hong Kong-based CK Life Sciences has announced the establishment of Sequencio Therapeutics, a wholly-owned sub

Guardant Health, Inc., a leading precision oncology company, has announced the launch of the Shield™ mul

BPL Medical Technologies, one of India’s leading medical technology companies, has recently announced th

Japan-based Fujifilm Corporation has announced its investment in Valanx Biotech, an Austrian biotechnology com

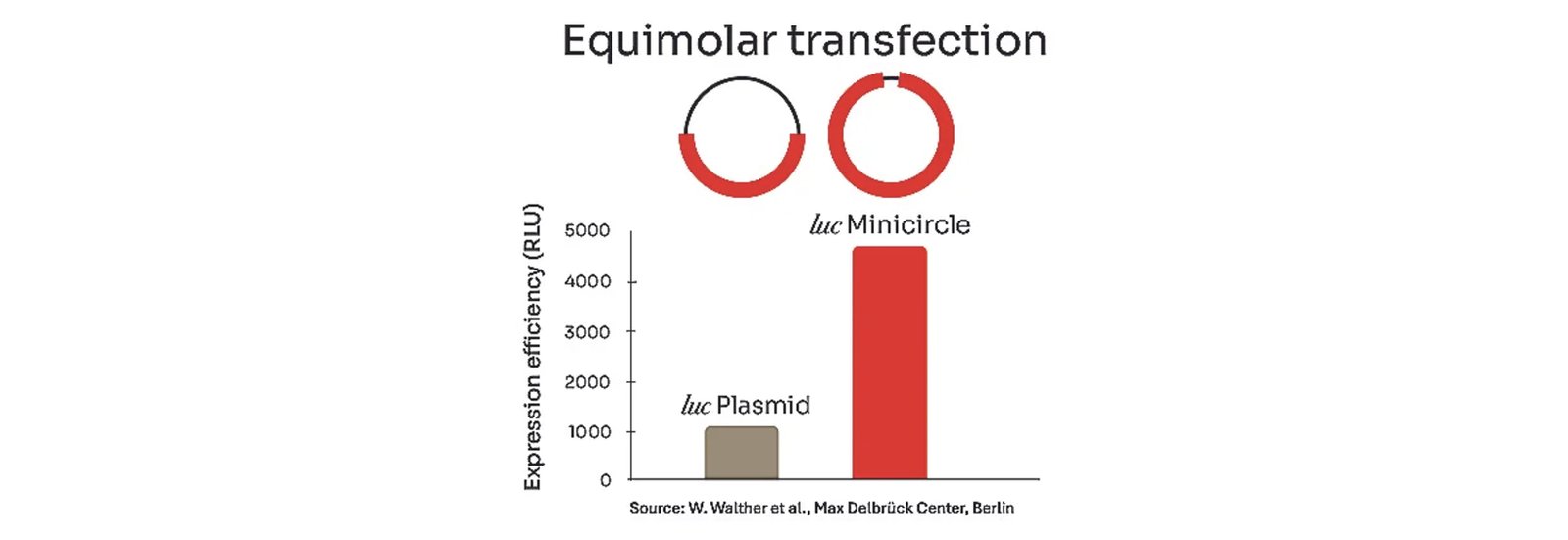
Australia-based startup BioCina, a global end-to-end Contract Development and Manufacturing Organisation

APAC Accelerates CERVICAL CANCER Elimination
Cervical cancer remains one of the most pressing women’s health challenges in the Asia Pacific region, accounting for approximately 58 per cent of global deaths, according to an Economist Impact analysis commissioned by the Asia Pacific Women’s Cancer Coalition and supported by Roche. Despite being largely preventable through vaccination and early detection, the disease continues to impose a substantial clinical, social, and economic burden across low, middle, and high income countries in the region. The World Health Organisation has therefore set a target to eliminate cervical cancer as a public health problem by 2030, defined as reducing incidence to fewer than 4 cases per 100,000 women. Achieving this goal requires meeting the 90 70 90 targets by 2030: 90 per cent of girls fully vaccinated against HPV by age 15, 70 per cent of women screened with a high performance test by ages 35 and 45, and 90 per cent of women with precancer or invasive cancer receiving appropriate treatment.
For Feedback, please email us at: communications@mmactiv.com
Dubai International Pharmaceutical & Technology Conference& Exhibition (DUPHAT)
Index Conferences & Exhibitions
info@duphat.ae https://duphat.ae/Dubai World Trade Center
ArabLab 2026 Dubai
Terrapinn Middle East Fz Llc
anne.deguzman@terrapinn.com https://www.terrapinn.com/exhibition/arablab-live/index.stmDubai World Trade Centre, Dubai
ArabLab 2026 Dubai
Terrapinn Middle East Fz Llc
anne.deguzman@terrapinn.com https://www.terrapinn.com/exhibition/arablab-live/index.stmDubai World Trade Centre, Dubai



