PATH welcomes the World Health Organization's (WHO's) prequalification of the thermostable rotavirus vaccine, ROTASIIL, manufactured by Serum Institute of India. The vaccine, which prevents severe rotavirus-induced diarrhea in infants, provides an innovative and affordable option to the global market as it is the first rotavirus vaccine that does not require constant refrigeration and will help meet the critical public health goal of improving vaccine supply worldwide.
Achieving WHO prequalification means that ROTASIIL will now be available for procurement by United Nations agencies and Gavi, the Vaccine Alliance, for use in low- and middle-income countries.
This will help accelerate availability of the vaccine in countries experiencing the highest burden of rotavirus-induced diarrhea by providing a practical and affordable option for vaccine introduction.
Rotavirus is the most common cause of severe diarrheal disease in children worldwide, and vaccination is the best way to prevent severe rotavirus illness. According to a recent study, 37 percent of global childhood diarrheal deaths in 2013 were due to rotavirus, a total of 215,000 rotavirus deaths. More than 90 percent of these deaths occurred in low-resource settings.
ROTASIIL was licensed by the Drugs Controller General of India in January 2017. Subsequently, data analysis from a Phase 3 study that gathered additional information required for WHO prequalification was completed and submitted in September 2017.
The study found no interference with the immune response to childhood vaccines when co-administered with ROTASIIL and confirmed the consistency of three different and independent lots of the vaccine produced by Serum Institute. India began using ROTASIIL in its national immunization program starting in April 2018 in Jharkhand state, with further roll-out expected. ROTASIIL is also currently available for sale in India's private market.
The international nonprofit PATH partnered with Serum Institute on evaluating ROTASIIL in the Phase 3 studies conducted in India and assisted with their applications for local licensure and WHO prequalification. PATH is continuing to work with Serum Institute on additional studies on ROTASIIL effectiveness (disease reduction in the population) and cost-effectiveness of the vaccine in India.
"PATH congratulates our partner, Serum Institute of India, for achieving the critical milestone for global access, WHO prequalification, of their oral rotavirus vaccine, ROTASIIL. PATH looks forward to contributing to the ongoing evaluation of this vaccine, and seeing a thermostable product added to the global supply of rotavirus vaccines in the fight against diarrheal disease," said Dr. David C. Kaslow, PATH's vice president for Essential Medicines and global head of the Center for Vaccine Innovation and Access.
Médecins Sans Frontières and Epicentre also evaluated the efficacy and safety of ROTASIIL in a separate Phase 3 study in Niger. The ROTASIIL used in the Niger study was stored at less than 25°C and transported for vaccination at ambient temperature, thus bypassing the typically challenging and costly cold-chain requirements that apply to most other vaccines. These data were included in the WHO prequalification dossier that led to the approval of the vaccine as a thermostable product.
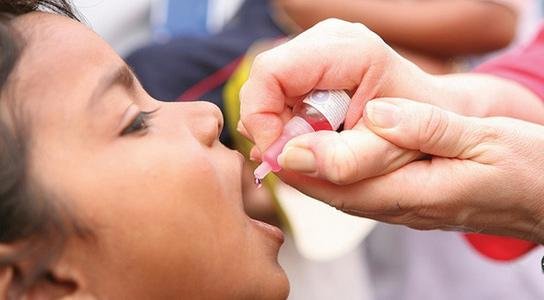
ROTASIIL is an oral vaccine administered to infants in a three-dose course at 6, 10, and 14 weeks of age, at the same time as routine vaccinations. The WHO-prequalified ROTASIIL formulation is a lyophilized (freeze-dried) presentation packaged in single-dose vials alongside a diluent. ROTASIIL contains bovine-human reassortant rotaviruses against the most common rotavirus serotypes (G1, G2, G3, G4, and G9).
The bovine-human reassortant rotaviruses were developed by the US National Institutes of Health and licensed to several emerging-country manufacturers and one US company for further development. Serum Institute was one of the licensees and developed it as ROTASIIL, a pentavalent (five-strain) vaccine product.




