Photo Credit: PRNewswire
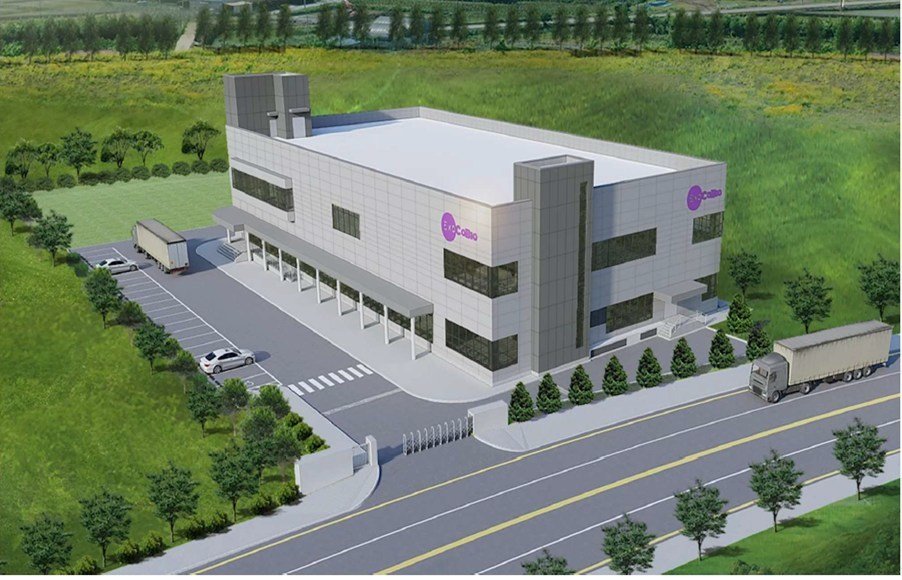
ExoCoBio Inc. has announced the initiation of its "Project EGMP", starting with the construction of the world's first exosome-based drug manufacturing & R&D facility (EGMP facility) in Osong, Chungcheongbuk-do, South Korea. Osong was created for a bio-cluster to attract biotechnology businesses with government agencies like Korea FDA.
ExoCoBio will invest approximately $20M to build the EGMP facility. The EGMP facility is expected to be completed by the end of 2022.
This EGMP facility, a two-story building with 21,000 sq ft per story on a site of 71,000 sqft, is expected to become a world-class facility of innovative exosome-based new drugs.
In particular, this EGMP facility will have installed two types of advanced exosome production lines, not only for naïve stem cell exosomes of regenerative medicine, but also for genetically engineered exosomes as a new future therapeutic portfolio, both of which have been continuously developed by ExoCoBio.
With the establishment of the EGMP facility, ExoCoBio's goal to apply for phase 1 clinical trials in the United States in 2023 is expected to gain stronger momentum for the development of a variety of exosome-based therapeutics against significant inflammatory diseases such as atopic dermatitis, SARS-CoV-2, Dupilumab Facial Redness (DFR), and others.
ExoCoBio is also planning to promote exosome CDMO business through partnering with global exosome researchers and companies who require large-scale exosome production.




