Photo Credit: Tokyo University of Science, Japan
The cells in our body are constantly fighting off the threat of cancer by repairing damaged DNA. In a new study, scientists from Tokyo University of Science investigate the structure of an elusive protein complex that plays a key role in the activation of the “Fanconi anemia pathway” involved in DNA repair, and report on the factors governing its stability. Their insights can potentially help find novel treatment disorders involving chromosomal instability, including cancer.
The process of DNA repair involves a complex interplay between several gene pathways and proteins. One such pathway is the “Fanconi anemia (FA) pathway,” whose genes participate in DNA repair. FANCM, a component of this pathway, is tasked with the elimination of harmful DNA “inter-strand cross-links,” and interacts with another component called MHF in order to function. The importance of the FANCM-MHF complex is well-documented: its loss can result in chromosomal instabilities that can lead to diseases such as FA itself and cancer. However, little is known about its structure and the basis of its stability.
Against this backdrop, Associate Professor Tatsuya Nishino and his colleague Dr. Sho Ito from Tokyo University of Science decided to explore the crystalline structure of this intriguing complex using X-ray diffraction techniques. “DNA damage and chromosome segregation are mechanisms necessary for the maintenance and inheritance of genes possessed by all organisms. MHF (also known as CENP-SX) is an enigmatic complex that plays a role in DNA repair and chromosome segregation. We wanted to find out how it performs these two different functions in the hope that it might give us insights into novel phenomena,” explains Prof. Nishino. Their findings are published in Acta Crystallographica Section F: Structural Biology Communications.
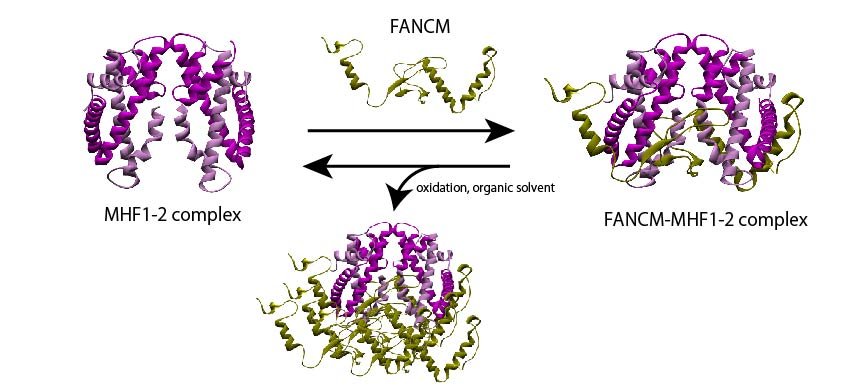
The scientists prepared a recombinant version of the FANCM-MHF complex, consisting of FANCM from chickens and MHF1 and MHF2. They were able to purify three different types of protein crystals—tetrahedral, needle-shaped, and rod-shaped—from similar crystallization conditions. Surprisingly, upon determining the structure with X-ray crystallography, they found that two of the crystal forms (tetrahedral and needle-shaped) contained only the MHF complex without FANCM.
Scientists examine the presence of a compound called 2-methyl-2, 4-pentanediol (MPD) caused the FANCM-MHF complex to disassemble. It is an organic solvent commonly used in crystallography, and exposure to an oxidizing environment.
The findings are extraordinary and can be used to improve the stability of the FANCM-MHF complex for future studies on its structure and function. Dr. Ito believes we have much to expect in the future from this complex. “A good understanding of this complex can help us treat cancer and genetic diseases, create artificial chromosomes, and even develop new biotechnological tools,” he speculates.




