Allergan plc announces the launch of Ozurdex® (dexamethasone intravitreal implant 0.7 mg) in China for the treatment of adult patients with macular edema secondary to retinal vein occlusion (RVO).
RVO is the second most common type of retinal vascular disorder after diabetic retinal disease and it is estimated that 7.4 million people in China are living with the condition.
RVO is caused by blockages in the central retinal vein (CRVO) or in the surrounding branch retinal veins (BRVO) which leads to disrupted blood flow, haemorraghing and eventually macular edema. If left untreated, RVO can result in sudden and painless loss of vision.vi Symptoms of the disease include blurry vision and floaters (dark spots and lines in vision).
Ozurdex® is the first approved intravitreal injection for RVO in China and is licensed for the treatment of macular edema following BRVO and CRVO.
Allergan has accelerated access to this treatment in 27 hospitals across China, making it available as a first-line treatment for this potentially sight-threatening condition.
Ozurdex® was put on the priority approval list by the Chinese Food and Drug Administration (CFDA) and was granted an Imported Drugs Licence (IDL) in October 2017.
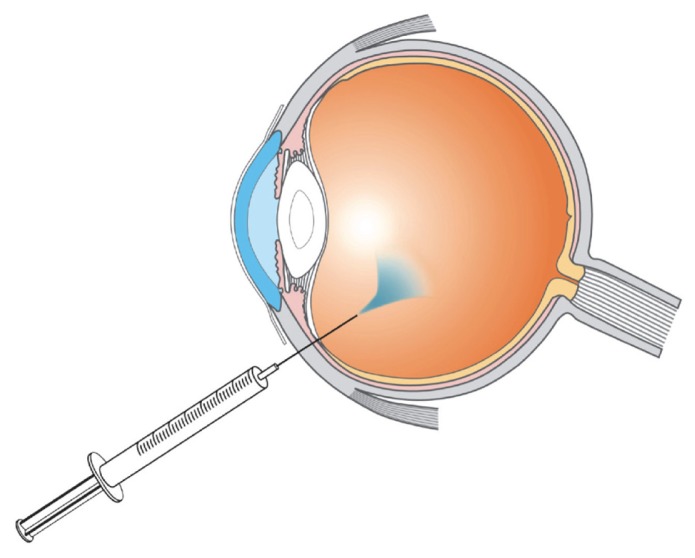
Ozurdex® is a biodegradable implant containing the anti-inflammatory corticosteroid, dexamethasone. It is implanted in the back of the eye, via a single use applicator, and the implant slowly releases dexamethasone directly to the retina over a period of several months. This treatment works by suppressing inflammatory mediators and delivers sustained visual gains.




