
Mr David Hoey, CEO, Vaxxas
Singapore: Though vaccination is considered the most important procedure for healthcare and disease management, it is not widely accepted by all sections of the society. Vaccination can be a painful procedure. Kids do not like them and many parents do not like seeing their children suffer. For most children, this important procedure may cause unnecessary stress, anxiety, and may also result in non-adherence to vaccination schedules. Developing a painless vaccine delivery method will help improve vaccine acceptance, completion of vaccine schedules and aid in improving health outcomes.
Located in Brisbane, Australia, Vaxxas is a biotechnology startup that aims at revolutionizing the current method of vaccination and has developed a needle-free vaccination procedure. Called the Nanopatch, the technology was invented at the University of Queensland's Australian Institute of Bioengineering and Nanotechnology. Nanopatch seeks to replace the traditional needle and syringe methods with a small patch that delivers vaccines painlessly directly to the body's immune cells.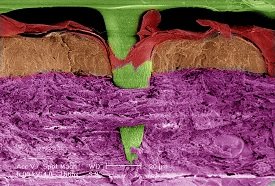
The Nanopatch technology is being advanced and commercialized by Vaxxas, a privately-held biotechnology company focused on enhancing the performance of existing and next-generation vaccines. The company was founded in August 2011 with the completion of a A$15 million [$12 million] Series A equity financing led by OneVentures with co-investors Brandon Capital, the Medical Research Commercialization Fund (MRCF), and the US-based HealthCare Ventures.
Vaxxas' Nanopatch contains an ultra-high density array of projections - invisible to the naked human eye - that are dry-coated with the vaccine. These projections directly deliver the vaccine to the immune cells below the outer surface of the skin, thus avoiding nerve endings that cause pain.
"Needle and syringe is by far the most dominant form of vaccination - for both systemic delivery as well as intradermal injection. The Nanopatch is fundamentally different, delivering a dry coating of the vaccine to the immune cells immediately under the surface of the skin. This ensures painless and highly efficient delivery of the vaccine," noted Dr David Hoey, CEO, Vaxxas.
Vaxxas claims that Nanopatch can potentially enhance the immune response with as less as 1/100 of the dose. "In a wide range of preclinical studies, Nanopatch delivery of a vaccine achieves equivalent protective immunity as the needle and syringe but with only 1/100th of the dose," explained Dr Hoey.
Vaxxas' method of vaccine delivery is also economically viable and will help in cutting down costs as the material used to coat the vaccine is temperature resistant and does not need specialized storage conditions. As a result, it strives to show that the traditional and costly cold-distribution chain is unnecessary and vaccines can be administered in parts of the world where cold-chain infrastructure not available.
"It has also been shown that Vaxxas' proprietary dry-coating technology can eliminate the need for vaccine refrigeration during storage and transportation - removing the resource burden of maintaining the cold chain," said Dr Hoey.
Leveraging both the potent immunogenic response and thermo stability, Vaxxas is applying its technology to improve the performance of vaccines, with initial applications targeting infectious disease and oncology. "The ultra-high density array that results in potent immunogenic response is unique to the Nanopatch. We believe that Nanopatch can be a more efficient and effective way to deliver vaccines for patients and for the industry," observed Dr Hoey.
Nanopatch is in its late stage preclinical studies and the first human trials are expected to kick off by late 2015. The technology has been demonstrated to deliver wide range of vaccines. "In preclinical studies the Nanopatch has shown promise in delivering a vast range of vaccines including DNA vaccines, VLPs, as well as split and inactivated viruses for a broad range of diseases," added Dr Hoey.
Moving ahead, Vaxxas is pursuing strategic plans to license its technology to global pharmaceutical companies as well as advance vaccine candidates on its own. The company has struck a deal with German-based Merck to evaluate, develop and commercialize Vaxxas' Nanopatch vaccine delivery platform for undisclosed vaccine candidates developed by Merck.
Dr Hoey concluded, "Vaxxas is initially focusing clinical development efforts toward the US and Europe. The company is examining opportunities in the large emerging APAC markets, particularly where Nanopatch features such as no requirement for cold-chain could be valuable differentiators. We have had great success working in preclinical models, now we need to translate the benefits of the Nanopatch to humans. We need to stay focused."




